The foam and its applications
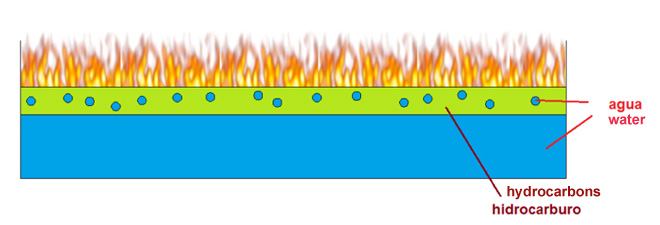
Hydrocarbons such as gasoline or diesel, have a lower surface tension than water. In case of fire and applying water alone, this and the hydrocarbon are mixed and rapidly the two fluids are separated, the hydrocarbon remains in the top and water at the bottom, being still in contact with the hydrocarbon and oxygen nullifying any suppression action by the water.
Action of water on hydrocarbon
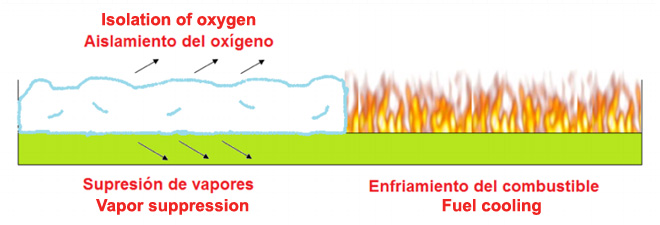
When the foam is mixed with water and air, the surface tension is reduced allowing the foam to float on the surface of the hydrocarbon. The presence of flammable vapors suppresses foam separating the oxygen and hydrocarbon ignition source, as well as by cooling the fuel below its ignition temperature.
Application of foam over the burning of hydrocarbon
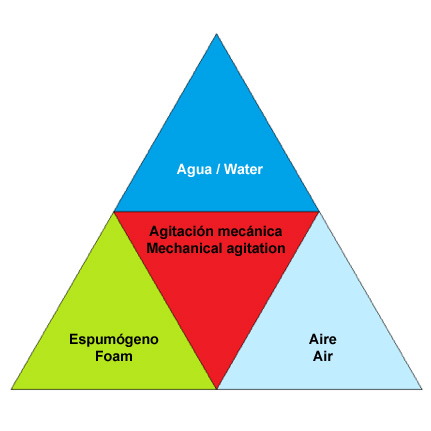
The foam is a mixture of foam, water, gas, usually air, and some mechanical agitation that when combined in the right proportions produce air-filled bubbles suffocate, insulate and cool fires of class A and B. These 4 elements represent tetrahedron foam. If any item is missing or is incorrect proportions, foaming may be affected.
Tetrahedron foam
The main properties of the foam are:
· Fluency, great ability to cover an area quickly.
· Heat resistance, resistance to thermal decomposition
· High cohesion of the same, with consequent elimination of vapors.
· Fuel tolerance, the ability of bubbles to resist adhesion of the fuel, when the foam is fed subsurface injected or from the top.
· High cooling ability due to its high water content.
· Slow drainage, plenty of water retention.
· Less dense than water and fuel and flammable liquid.
The main applications for fire protection foam are:
| Industry | Application | Usual type foam* | Petrochemistry | Storage tanks for flammable liquids | Low expansion | Petrochemistry | Retention ponds and / or collection | Low and medium expansion | Petrochemistry | Process areas | Low, medium and high expansion | Petrochemistry | Docks, marine loading docks | Low and medium expansion | Petrochemistry | Fuel racking areas | Low expansion | Petrochemistry | Rafts GNL and GLP | Medium and high expansion | Petrochemistry | Process areas | Low, medium and high expansion | Chemistry | Fuel Storage “A” and “B” | Low and high expansion | Aeronautics | Aircraft hangars | Low and high expansion | Aeronautics | Heliports | Low and high expansion | Electrical | Transformers | Low, medium and high expansion | Electrical | Cable tunnels | High expansion |
*types of foam are discussed in the following section.
Protection systems are not suitable for foaming, among others, the following applications:
· Chemicals, such as cellulose nitrate, that release oxygen or other oxidizing agents that can support combustion.
· Electrical fires.
· Metals such as sodium or potassium and sodium-potassium alloy in contact with water produce exothermic reactions.
· Water reactive Hazardous materials such as triethyl aluminum and phosphorus pentoxide.
· Metal fuels such as aluminum and magnesium.
Bibliography:
– Fire Service Pump Operator: Principles and Practice, 2011. International Association of Fire Chiefs National
Fire Protection Association.
– Presentación J.R. CARME LUESMA, Noviembre 2003.
– UNE-EN-13565-2. Sistemas fijos de lucha contra incendios. Sistemas espumantes. Parte 2: Diseño, construcción y mantenimiento.





